.avif)
.avif)
For Molecular Diagnostics Companies
to first data delivery
for model selection queries
reduction in manual data requests











Valuable real-world genomic data from thousands of patients remains undermonetized due to lack of efficient pharma licensing infrastructure.
.svg)
Each pharma partnership requires 6-12 months of legal negotiations and custom technical infrastructure, severely limiting deal velocity.

Internal R&D teams and external partners need access to the same multimodal clinical testing data, but disconnected systems force you to manage everything separately—slowing down both.


"Manifold helps us prioritize speed and specialization. With their AI capabilities combined with our proprietary multi-modal data, FoundationInsights® powered by Manifold is resonating with customers and partners.”
Brian Clancy
Senior International Product Manager, Data Solutions & Partnerships, Foundation Medicine
Explore Manifold's core solutions
.avif)
.png)
Fit-for-purpose solutions for clinical research leaders in cancer and rare disease.
Streamline legacy systems and manual processes with a single study and data management platform.
Reduce and turnaround time and increase productivity by connecting biospecimen data and clinical data in a single data management platform.
Unify clinical and multimodal data on a modern platform to enable automated data preparation into a research-ready patient model.
Answer research questions about diagnoses, treatments, and outcomes quickly and easily.
Bring multimodal data together so your teams can make faster pipeline decisions, design prospective studies, and identify biomarkers.
.svg)
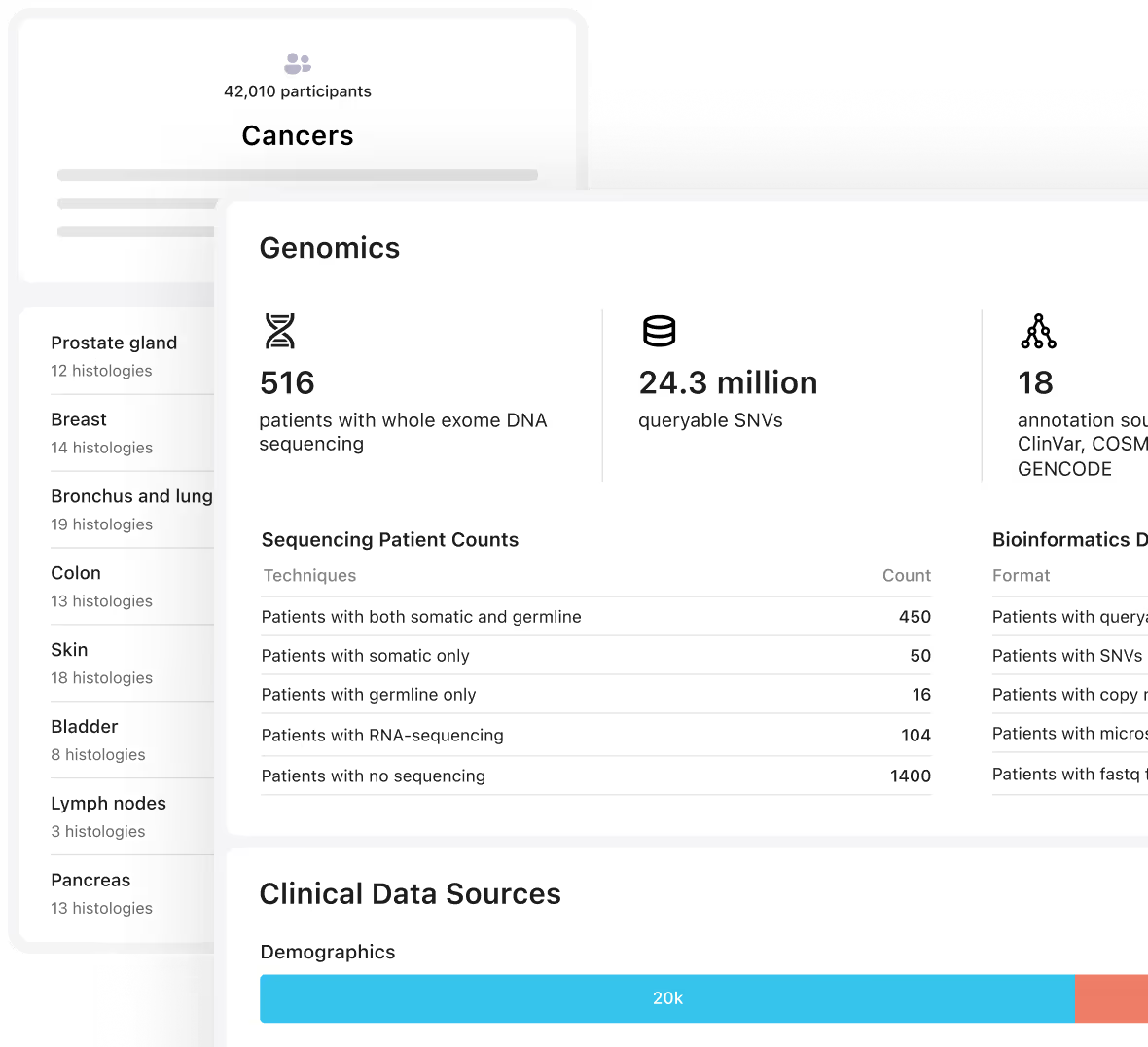
Unified access to genomic, clinical, and outcomes data
.svg)
AI-assisted data exploration and analysis
.svg)
Self-service access for bioinformatics teams
Molecular diagnostics companies using platform for both internal R&D and external commercial partnerships
Secure, compliant, auditable data access that meets pharma ITand legal requirements out of the box
.svg)
100% audit trail for all data access
.svg)
Tiered access controls and permissions
.svg)
Automated usage metering and billing
.svg)
Pre-configured for pharma IT requirements
Leading CRO serving multiple pharma partners simultaneously through single infrastructure with tiered access controls
Serve internal R&D teams and external partners through a single platform — no incremental engineering required.
.svg)
Onboard new partners in days, not months
.svg)
Role-based access for internal teams and external collaborators.
.svg)
Central management dashboard for all partnerships
.svg)
Unified view across multimodal datasets.
One organization serves multiple pharma partners simultaneously, with self-service reducing manual data delivery requests by 50%+
Go from first conversation to data delivery in 3-4 months, not 12-18 months
.svg)
Pre-negotiated template agreements reduce legal cycles
.svg)
Pharma-approved security & compliance out of the box
.svg)
No custom engineering required for each partnership
One molecular diagnostics company reduced partnership setup time from days to minutes for model selection queries

Unify multimodal data to accelerate pipeline decisions and biomarker prioritization
Design and execute prospective studies with governed access to historical testing data
License real-world genomic data for target discovery and biomarker identification
Enable CDx development with real-world validation datasets
Support regulatory submissions with longitudinal patient outcomes
Power patient identification and recruitment services
Deploy in 3-4 months at a fraction of the cost. No multi-year engineering projects or maintenance fees required.
Active pharma partnership pipeline or strategic priority to modernize how teams access clinical testing data
Want to serve 3+ pharma partners without rebuilding infrastructure each time
Need to reduce partnership setup time from 12+ months to 3-4 months
Need enterprise-grade security, audit trails, and governance for pharma partnerships
.svg)
Setup complete in 3-4 months
.svg)
Support multiple collaborators simultaneously
.svg)
Enterprise-grade security & compliance
.svg)
Flexible access models