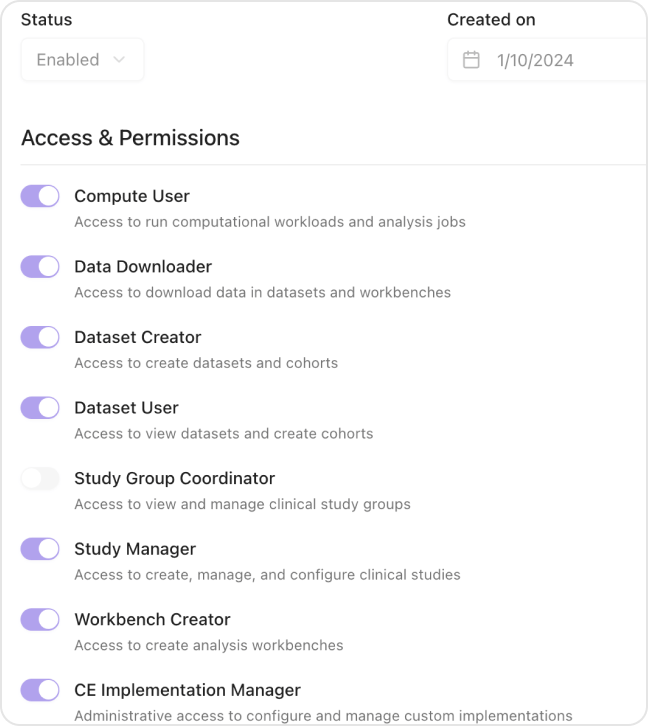
Comprehensive privacy, consent, and compliance frameworks that meet pharma requirements
.svg)
Patient privacy and consent management
.svg)
100% audit trail for all data access
.svg)
Role-based access controls
.svg)
Automated compliance reporting
Leading organizations manage governed data access for multiple pharma partners through single platform
Reduce partnership setup from months to weeks with pre-built frameworks
.svg)
Pre-negotiated template agreements
.svg)
Pharma-approved security architecture
.svg)
No custom engineering per partner
Organizations reduce partnership setup time significantly through standardized infrastructure
Serve multiple biopharma partners simultaneously without rebuilding infrastructure each time
.svg)
Onboard new partners in days
.svg)
Partner-specific workspaces and permissions
.svg)
Automated usage tracking and billing
Leading organizations serve multiple pharma partners simultaneously through single infrastructure
.svg)
.svg)
.svg)
.svg)











.svg)
.svg)
.avif)
.png)